
As it is clear entropy depends on the randomness of the particles of the system so when there is more randomness of particle entropy will be positive and if the randomness of particles is less entropy will be negative only because this reason entropy of gases is greatest because there is more movement of particles as compared to other states of matter, followed by liquid and then least in solids therefore for all cases where transformation is from solid to liquid(melting of ice), solid to gas, liquid to gas(vaporization) entropy will be positive and for the cases when transformation is from gas to solid(conversion of carbon dioxide to dry ice) or liquid(condensation), entropy would be negative.Įnthalpy is the measure of complete energy of a thermodynamic system that includes all the transfers of energy in the system, it depends on the region of the system and for a homogeneous system it is sum of internal energy of the system and the product of volume and pressure hence it can be clearly observed that it depends on the size of system. 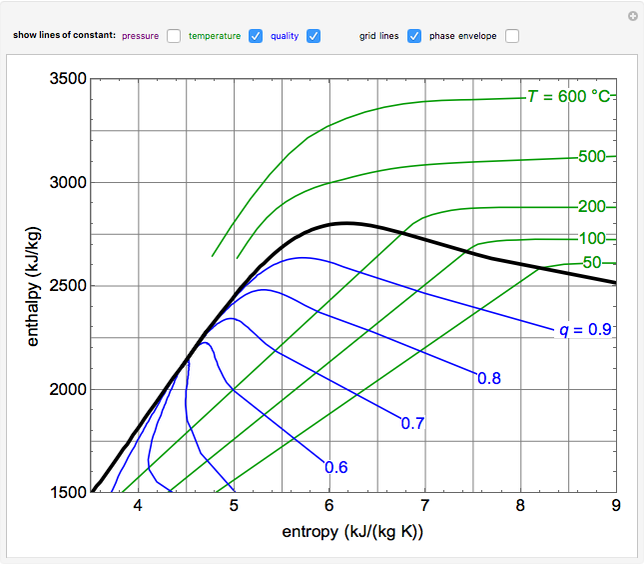
The value of entropy is positive and negative as well depending on the case. 
To understand it better let me give you an example suppose you place hot cup of tea in the refrigerator then you closed its door then surrounding of cup will absorbs heat from it until they both comes to the same temperature. Entropy can be the heat absorbed in a system which is reversible and the work did is a part of it. When we plot a graph between entropy and enthalpy at an absolute temperature, we will get a linear graph.Įntropy word is taken from Greek word that means transformation, it is a property of thermodynamic system(a system or region which is expressed with the help of its thermodynamic quantities like volume, density, temperature etc). Thus, entropy and enthalpy are directly proportional but always remember that from second law of thermodynamics, the entropy change of a closed system can never be negative or decrease. This relation signifies that when ever there is an increase in the total thermal energy(enthalpy) of the system, the degree of disorder (i.e.entropy) of the system increases. The relationship between enthalpy and entropy of a closed system is given by : This means, in a hypothetical reversible process(like carnot engine), entropy may remain constant and in an irreversible system the entropy will increase but in any closed system you will not find any decrease in the entropy. In other words, it is actually the heat or thermal energy that cannot be used to do work by the system and thus it represents the randomness of the particles.Īccording to second law of thermodynamics, the entropy of a closed system never decreases. There is a direct relation between enthalpy change, work done and heat transferred from and to the thermodynamic system.Įnthalpy can also be considered as a 'Heat content' of a system under constant pressure condition.Įntropy is the measure of disorder of a closed thermodynamic system. It keeps on changing whenever a system interacts with its surrounding. 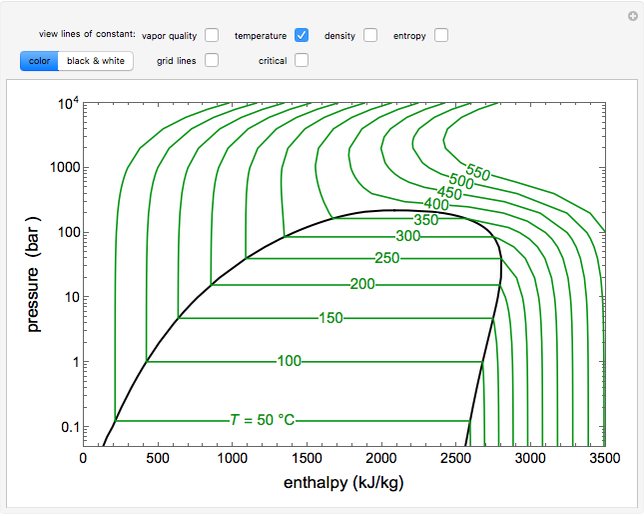
When ever a work is done by the system, there is a decrease in its entropy and when ever some work is done on the system, energy is gained and thus its entropy increases. The term Enthalpy in a thermodynamic system represents its total internal energy.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
