Furthermore, if stored in sufficient quantities, ammonia is considered corrosive as well as harmful. It is a common nitrogenous waste of aquatic creatures and an important component of terrestrial species' nutritional requirements. 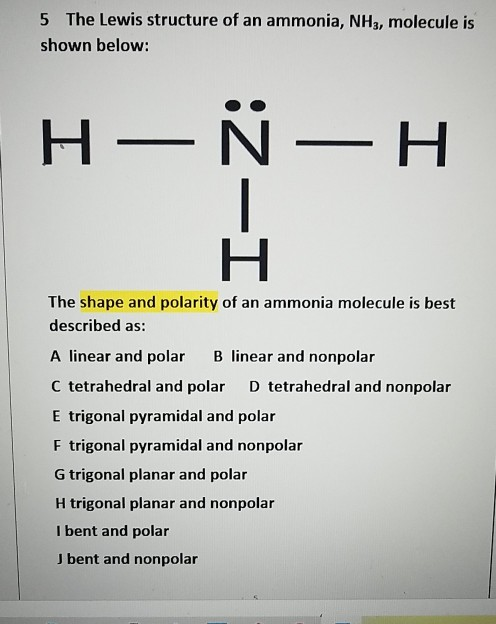
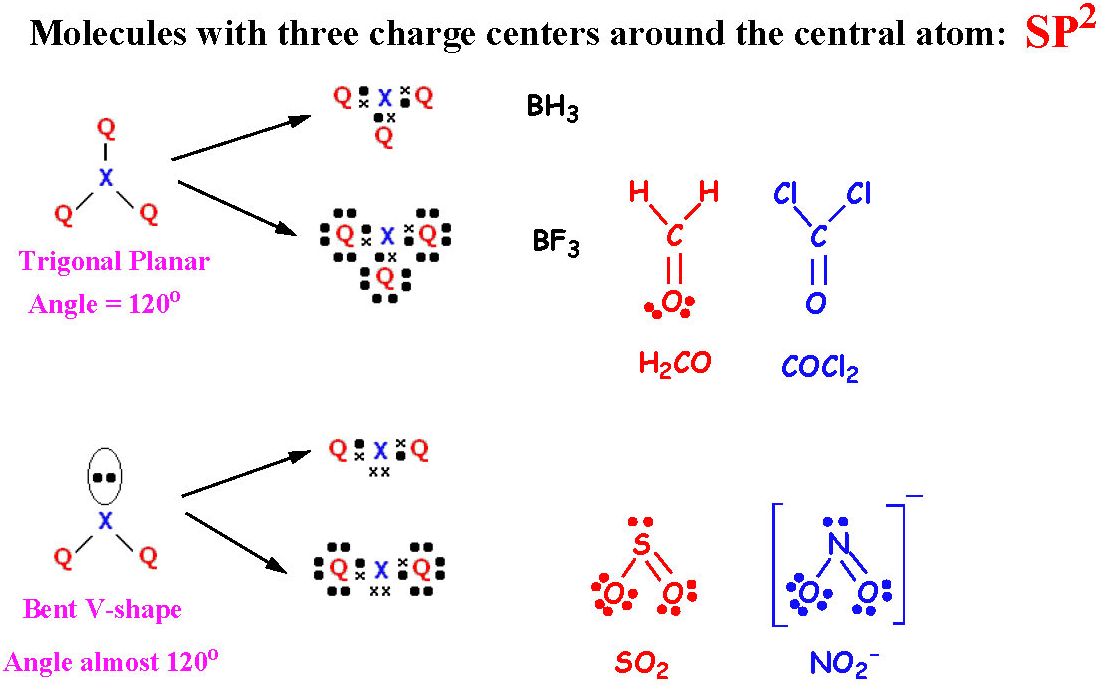
The fourth sp 3 orbital, on the other hand, is a nonbonding pair of hybridized orbitals that is generally employed to hold the lone pair together. One 2s orbital and three 2p orbitals of nitrogen mix during the production of ammonia to generate four hybrid orbitals with equivalent energy, which is referred to as an sp 3 kind of hybridization.įurthermore, if we look at the NH 3 molecule, we can see that nitrogen's three half-filled sp 3 orbitals create bonds with the three hydrogen atoms. The atomic number of nitrogen is 7, and its ground state is 1s 2, 2s 2 ,2p 3 according to its atomic number. To comprehend ammonia hybridization, we must investigate the surroundings surrounding Nitrogen. The electron geometry and molecular geometry are the same if all the electron groups are bonded and there are no lone pairs. When an electron that is not connected to a lone pair of electrons or another atom is found in the molecule, the shape of the molecule changes, not the shape of the electron. The arrangement of electron groups is called electronic geometry. Hybridization was proposed as the best explanation for why all C - H bonds in molecules like methane are identical. The new, hybridised orbital characteristics and energy are an 'average' of the original unhybridized orbitals. The new orbital can accommodate the same number of electrons as the old ones. When atomic orbitals combine to generate a new atomic orbital, this is known as hybridization. Lone pairs of excess electrons are shown as pairs of dots next to the atoms.Īlthough main group elements in the second period and beyond normally react by acquiring, losing, or sharing electrons until they have a complete octet of (8) electrons in their valence shell electron configuration, hydrogen (H) can only form bonds that share two electrons. Between atoms that are linked together, lines are drawn (pairs of dots can be used instead of lines). Lewis structures use chemical symbols to represent each atom and its place in the molecule's structure. Lewis structures add lines between atoms to represent shared pairs in a chemical bond, extending the concept of the electron dot diagram. Lewis named the Lewis structure after himself after introducing it in his 1916 article The Atom and the Molecule. All covalently bonded molecules and coordination compounds can be represented by the Lewis structural formula. Lewis structures, also known as Lewis point structures, Lewis point structures, electron point structures, or Lewis electron point structures (LEDSs), contain bonds between atoms within a molecule and all lone pairs that can be gifts. This section post will cover the Lewis dot structure, electron geometry, and molecular geometry of this molecule. Accepting a proton allows it to become an NH 4 + ion. It's a stable hydride with one nitrogen and three hydrogen atoms in it. We'll look at how to figure out if NH 3 is hybridized in this article.Īmmonia is a colourless chemical that is utilized in fertilizer production. 
Ammonia (NH 3 ) is sp 3 hybridized, or to be more specific, the central atom of ammonia, nitrogen.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
